200 g of a sample of limestone liberates 66 g of CO2 on heating. The percentage purity of CaCO3 in the limestone is Options:a 95
5 (489) · $ 5.50 · In stock
200 g of a sample of limestone liberates 66 g of CO2 on heating. The percentage purity of CaCO3 in the limestone is Options:a 95
200 g of a sample of limestone liberates 66 g of CO2 on heating- The percentage purity of CaCO3 in the limestone is Options-a- 95

Applied Sciences, Free Full-Text

Chemistry Class 10 part 1

PhEd-Some Basic Concepts of Chemistry-W.S, PDF, Mole (Unit)

Qdoc - Tips Chemical Engineering Reviewerdocx, PDF, Gases

Thermodynamics: Synopsis, PDF, Solvation

Basic Chemical Calculations-Merged, PDF, Mole (Unit)
When a limestone of mass 150g was heated until it decomposed to CaO, only 63g of CaO were obtained. What is the percentage purity of the limestone? - Quora
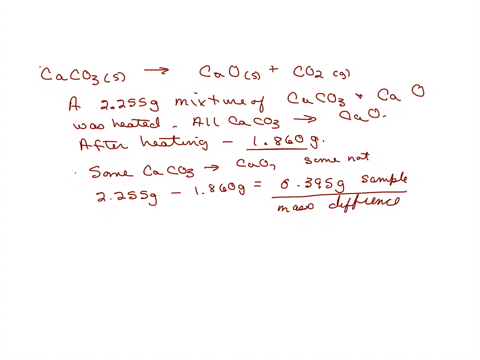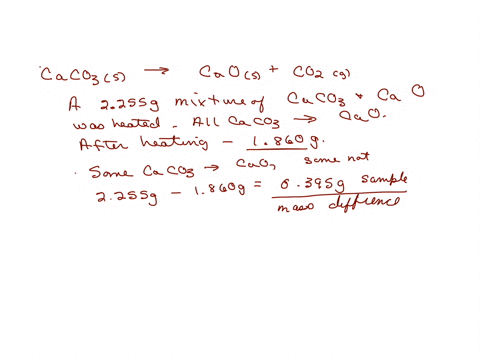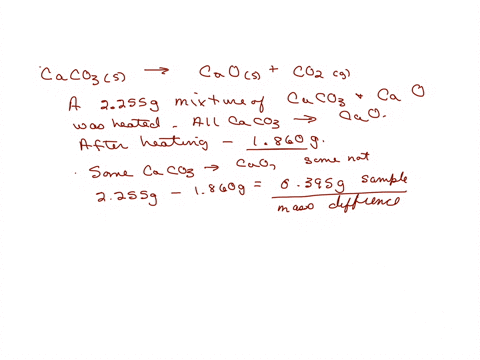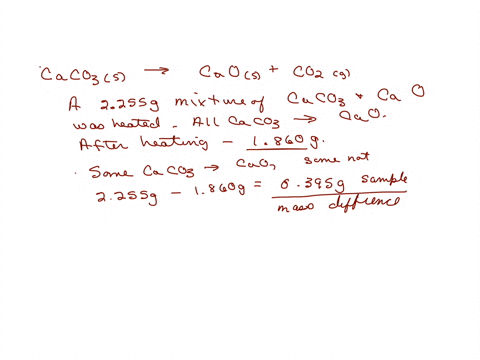
SOLVED: The weight percentage of limestone is as follows: CaCO3 = 93%, MgCO3 = 5%, Insoluble = 2%. This limestone is burned in a vertical kiln to produce lime (CaO, MgO, Insolubles).
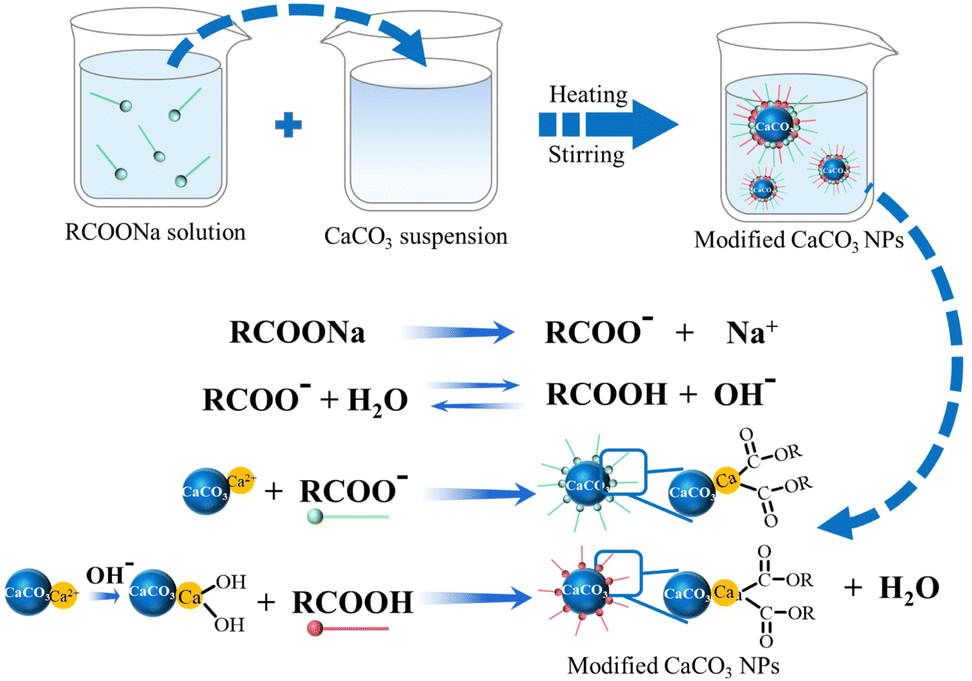
Calcium carbonate: controlled synthesis, surface functionalization, and nanostructured materials - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D1CS00519G

Solved A sample of limestone and other soil materials was

Chapter 10 Stoichiometry and Process Calculations

Unical Science 2o22