Cancer Biomarker Detection With Luminex Assays
5 (719) · $ 11.50 · In stock

Luminex and Bio-Techne are working together to support the development of early diagnostic solutions that detect cancer earlier through the use of proteomics.
Charles Rosser, a research scientist and professor of biomedical sciences, has developed a multiplex test for early-stage bladder cancer using Luminex® xMAP® Technology. Bladder cancer affects over 570,000 people globally each year, and the Oncuria® test aims to detect bladder cancer, monitors for recurrence, and predicts which patients will benefit from the immunotherapy treatment. Current diagnostic assays lack sensitivity and do not provide a comprehensive view of the cancer’s molecular profile, but the Oncuria® test covers 10 glycoproteins to detect the biological signature of bladder cancer with sensitivity of 90% to 93% and specificity of 86% to 95%. The test received Breakthrough Device Designation status from the FDA and is currently available as a laboratory-developed test (LDT). In a recent clinical validation study involving about 350 patients, the test offered 93% sensitivity. Learn more about this major shift in the care of patients with bladder cancer.

Bio-Techne on LinkedIn: Cancer Biomarker Detection With Luminex Assays

Quantum-dot-based suspension microarray for multiplex detection of lung cancer markers: preclinical validation and comparison with the Luminex xMAP® system

Cancer Biomarker Detection With Luminex Assays

Overview of Luminex® Multiplex Assay Technology

Hallmarks of Cancer ELISA Kits and Multiplex Immunoassays
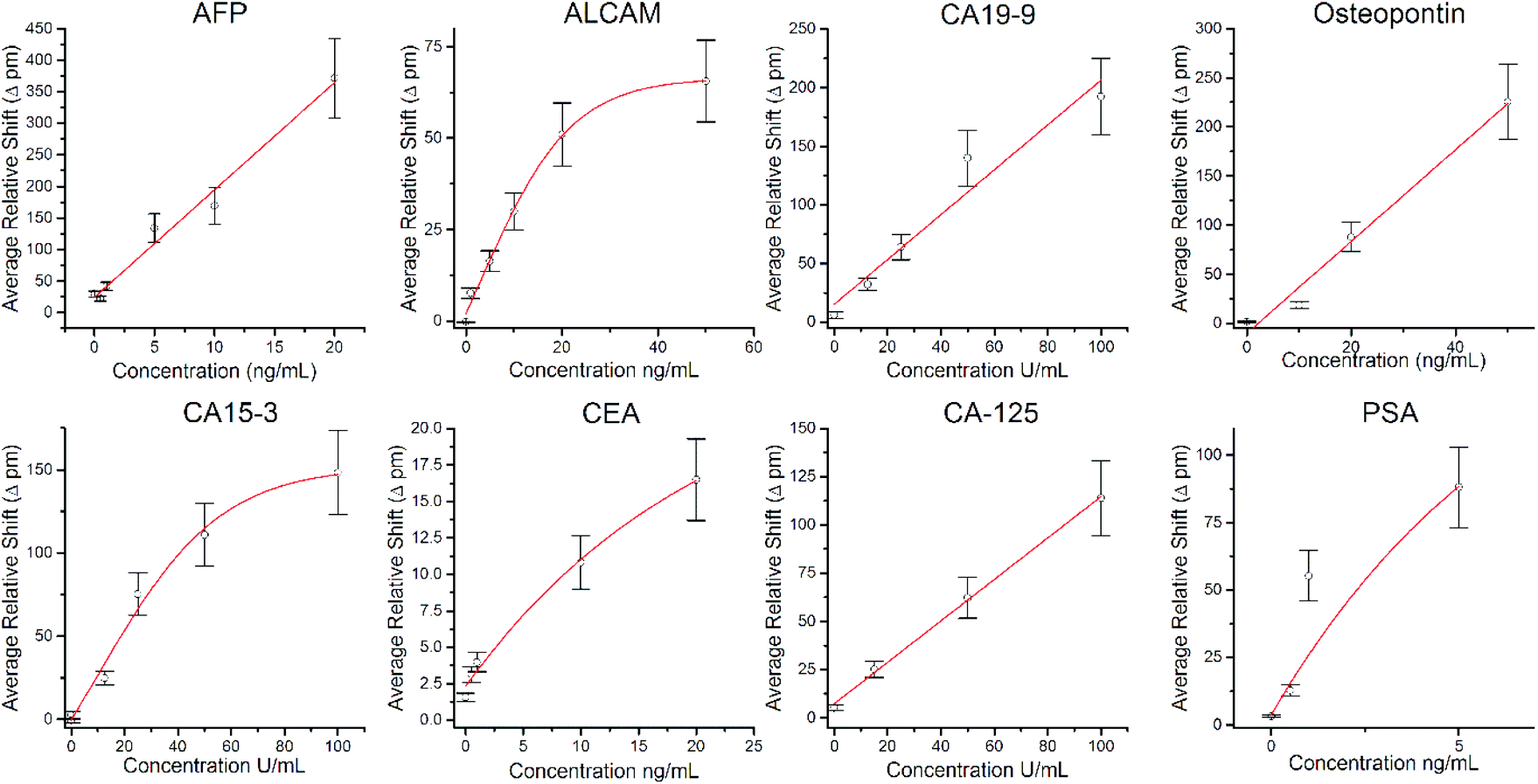
Multiplexed cancer biomarker detection using chip-integrated silicon photonic sensor arrays - Analyst (RSC Publishing) DOI:10.1039/C6AN01076H

Cancer Biomarker Detection With Luminex Assays
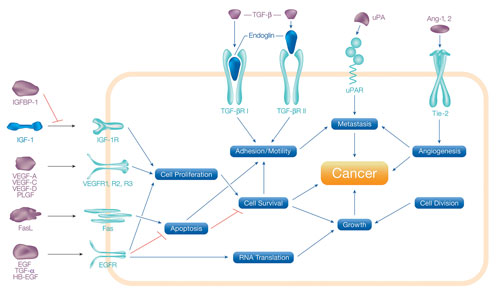
Multiplex Immunoassay for Analyzing Cancer Processes

Cancers, Free Full-Text
Biomarker Assays Irving Institute for Clinical and Translational Research
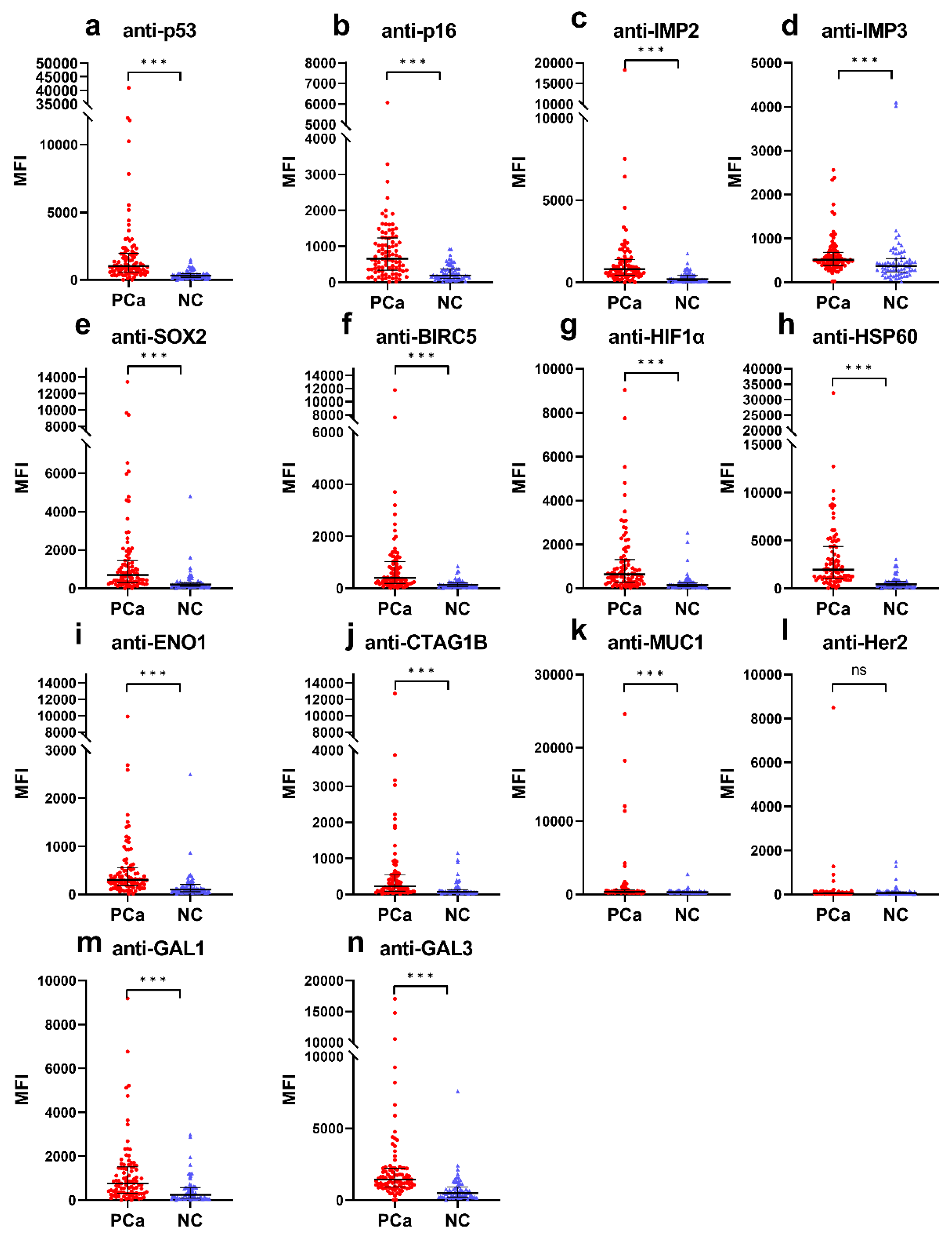
Cancers, Free Full-Text




/e9fb84c5-0bee-47d8-97a3-3c8098c747b1--sweet-and-spicy-pretzel-nut-mix_food52_mark_weinberg_14-11-18_0075.jpg)