Compressibility Factor, z vs Pressure, P (kPa), line chart made by Jdvani
4.7 (115) · $ 10.00 · In stock
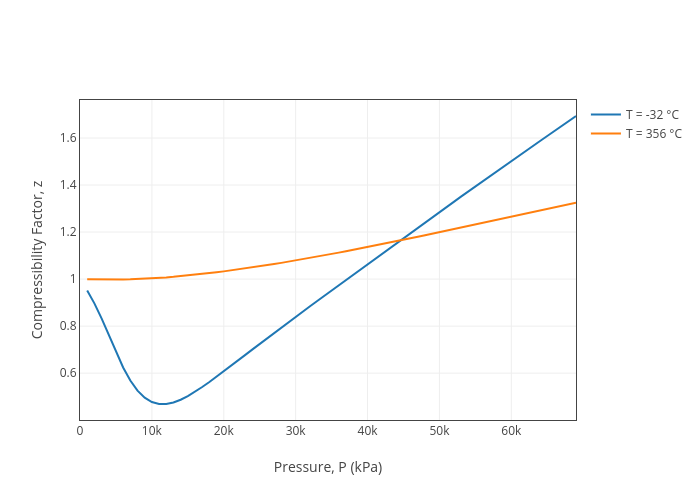
Jdvani's interactive graph and data of "Compressibility Factor, z vs Pressure, P (kPa)" is a line chart, showing T = -32 °C vs T = 356 °C; with Pressure, P (kPa) in the x-axis and Compressibility Factor, z in the y-axis..

The given graph represent the variations of Z (compressibility factor (Z)=dfrac {pV}{nRT}) versus P, three real gases A, B and C. Identify the only incorrect statement.For the gas B, b=0 and its

3.2 Real gas and compressibility factor – Introduction to Engineering Thermodynamics

Is z (compressibility factor) vs P (pressure) graph drawn by changing volume? If it is why it isn't drawn by changing mole - Quora

Compressibility factor - Wikipedia

Compressibility Chart: How does a Tr value of 1.8 correspond to 0.55 in Pr value for a SuperHeated Vapor. just confused because the Pr axis is spaced so weirdly. : r/EngineeringStudents

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange
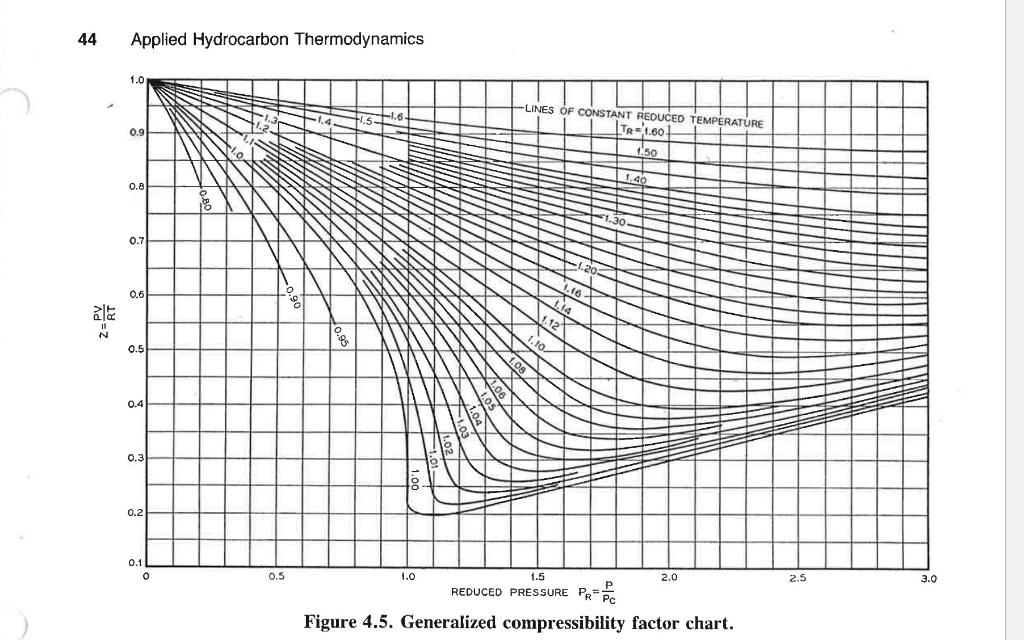
Solved Let us use the generalized compressibility factor

New compact Equations for the Compressibility Factor Z and Density of Liquid and Vapor Carbon Dioxide

The role of the compressibility factor Z in describing the volumetric behavior of gases

gas laws - How to find the temperature relationship between the isotherms in a compressibility factor (Z) vs pressure graph? - Chemistry Stack Exchange
Compressibility Factor and Compressibility Charts, Thermodynamics

Compressibility factor (gases) - Knowino

The given graph represents the variations of compressibility factor `Z=PV//nRT` vs `





