PDF] Hemoglobin polymorphism in white-tailed deer: subunit basis
4.9 (703) · $ 5.00 · In stock
![PDF] Hemoglobin polymorphism in white-tailed deer: subunit basis](https://d3i71xaburhd42.cloudfront.net/442de7651b90997b8b5c2ac0309ecaf101fed8da/4-Table1-1.png)
It was concluded from the results of limited structural studies that there were multiple peptide differences upon comparison of three non-α polypeptide chains in white-tailed deer. A variety of aberrant erythrocyte forms have been related to seven adult and two fetal hemoglobins in white-tailed deer. While sickling of the erythrocyte was not associated with a single hemoglobin type, it was precluded by hemoglobin V or VII, even when in combination with other hemoglobin types normally associated with sickling. The subunit basis of the hemoglobin polymorphism was presented. Two kinds of α subunits, six kinds of β subunits and one γ subunit were related to the whole hemoglobin molecule. The heterogeneity of the deer hemoglobins was based upon a variety of combinations of these numerous polypeptide chains. It was concluded from the results of limited structural studies that there were multiple peptide differences upon comparison of three non-α polypeptide chains.

Hematology SpringerLink

PDF) Gene Flow in the Face of Countervailing Selection: Adaptation

PDF) (TAAA)n-Alu Element Polymorphism in Vitamin D Binding Protein
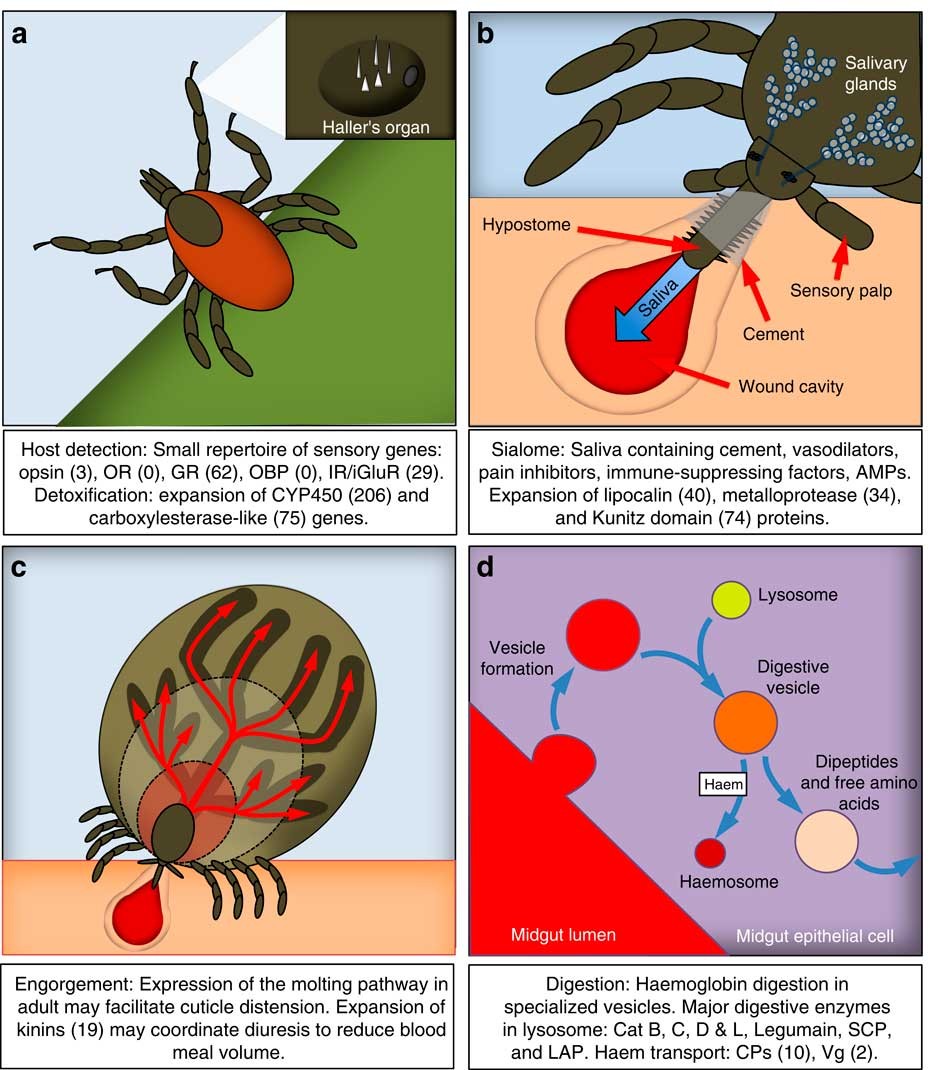
Genomic insights into the Ixodes scapularis tick vector of Lyme

PDF) Brief communication. Evidence of female heterogametic genetic

PDF) Hemoglobin polymorphisms affect the oxygen binding properties

Hemoglobin-oxygen affinity in high-altitude vertebrates: is there

PDF) Hemoglobin polymorphisms affect the oxygen binding properties

Multiplicity and Polymorphism of Fish Hemoglobins

The conserved Phe GH5 of importance for hemoglobin intersubunit

Fin whale MDH‐1 and MPI allozyme variation is not reflected in the

Animals, Free Full-Text







