ISPE GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized
4.9 (129) · $ 25.99 · In stock

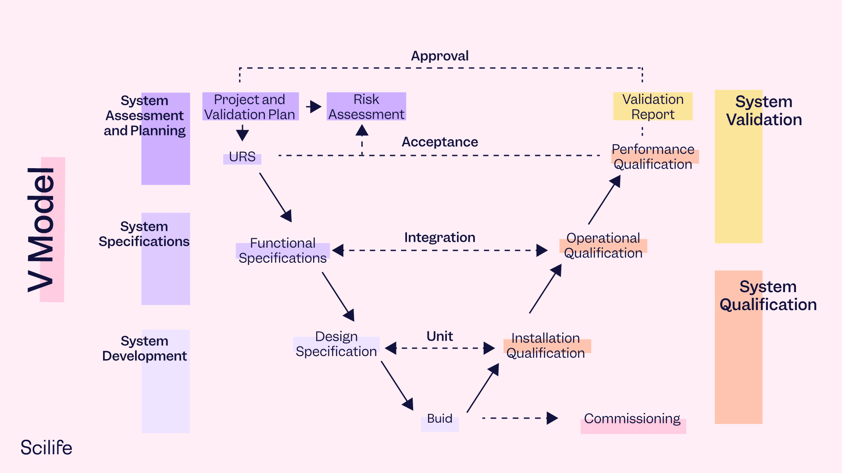
/hs-fs/hubfs/vmodel-v02.png?width=84

The Use of AI and ML in Biomanufacturing: Process and Product Validation Lifecycle and AI American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

PDF) GAMP 5 A Risk Based Approach to A Risk-Based Approach to Compliant GxP Compliant GxP Computerized Systems

Basic Principles of Computerized Systems Compliance GAMP 5 Online Training Course Demo

PDF) Whitepaper Gamp5 Risk Based Approach

GAMP 5 Guide 2nd Edition, ISPE

ISPE GAMP Good Practice Guide: A Risk-Based Approach to Regulated Mobile Applications

ISPE on LinkedIn: ISPE GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized Systems…

Achieving & Maintaining GAMP 5 Compliance: Risk-Based Approach to Software Development & Verification

RescopGxP (@RescopGxP) / X

Download ISPE GAMP 5: A Risk-Based Approach to Compliant GxP Computerized Systems, Second Edition In PDF - Standards Global

White Paper Gamp5 Risk Based Approach, PDF, Verification And Validation

ISPE - The much-anticipated ISPE GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized Systems (Second Edition) is now available! Maintaining the principles and framework of the first edition published in 2008
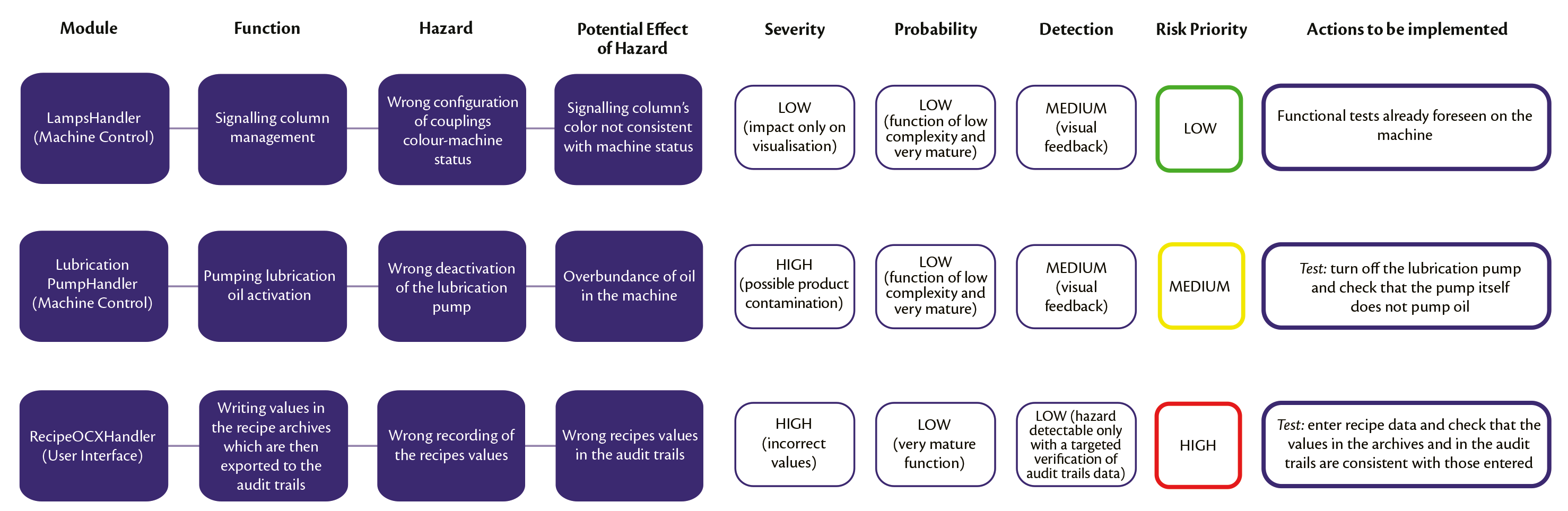
Achieving and maintaining GAMP® 5 compliance: IMA Active's risk-based approach to software development and verification • Pharma • IMA Group







