AIROS Medical Receives FDA Clearance to Market New Peristaltic
4.9 (146) · $ 14.99 · In stock

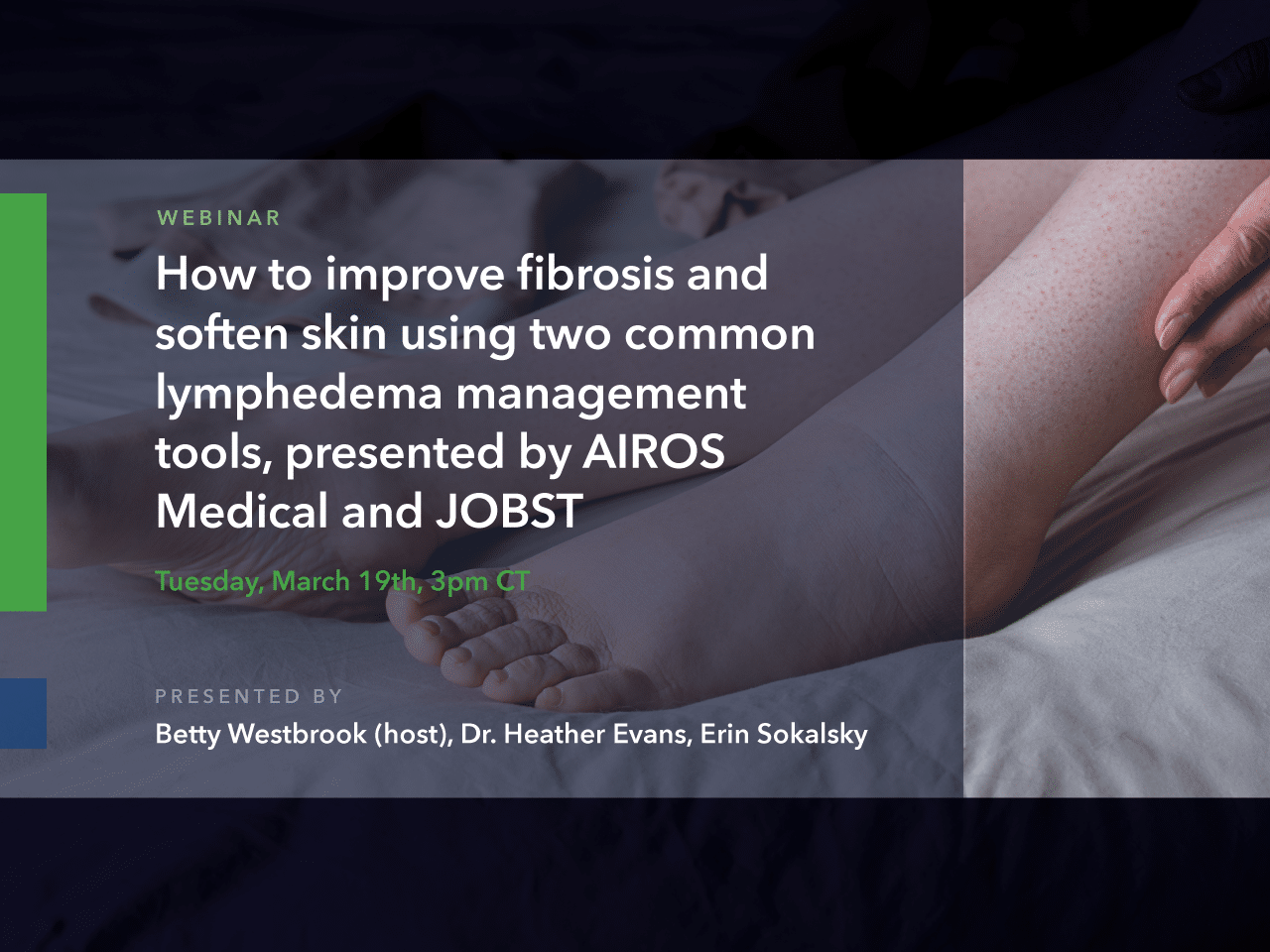
AIROS Medical announces FDA 510k clearance to market the AIROS 8P compression device and Pants garment that treats leg and pelvic swelling.

Cardio Flow, Inc., Announces FDA Clearance for FreedomFlow Peripheral Guidewire, plus First Commercial Case Completed in U.S. - Cardio Flow, Inc.
Articles & News - Page 2 of 7 - AIROS Medical, Inc.

Latest Health News in America
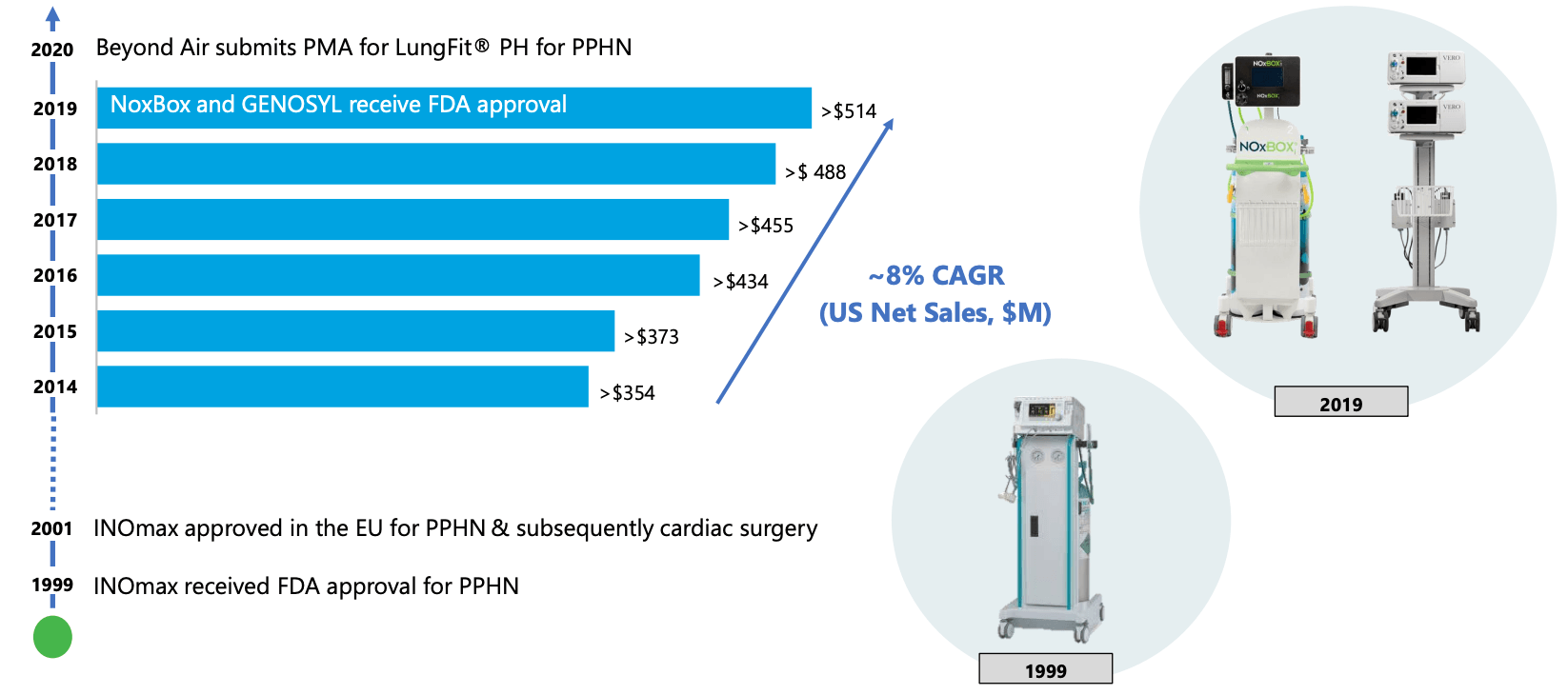
Beyond Air Stock: Tiny Company With Truly Massive Potential (NASDAQ:XAIR)

AIROS Medical (@AirosMedical) / X

AIROS Medical (@AirosMedical) / X

AIROS Medical Granted U.S. Trademark Registration for Company, Brand Name

Navy Removal Scout 800 Pink Pill Assasin Expo Van Travel Bothell Punishment Shred Norelco District Ditch Required Anyhow, PDF

AIROS Medical Receives FDA Clearance to Market New Peristaltic Pneumatic Compression Device, Truncal Garments for Lymphedema Treatment

MyChesCo's Regional News Southeastern Pennsylvania News- Page 456 of 563

Hei-VAP Industrial Rotary Evaporators, Heidolph®

FDA approves LungFit PH to treat respiratory failure in neonates
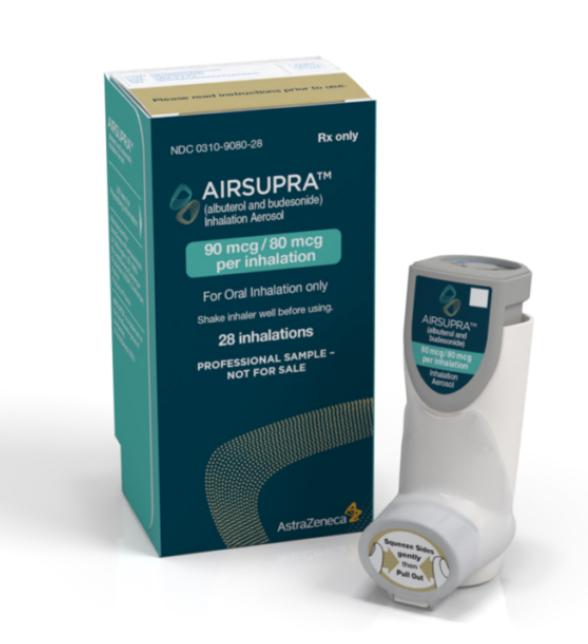
Airsupra (albuterol and budesonide) FDA Approval History

Compression Therapy Device Technology for Lymphedema Treatment
Darren Behuniak, Author at AIROS Medical, Inc.







