Developing a Thermodynamical Method for Prediction of Activity
5 (144) · $ 19.50 · In stock

Results of the experimental measurements on the partial molar volume of kerosene used as a medium for dissolving TBP are utilized to determine the activity of TBP in the binary kerosene-TBP solution through the application of Gibbs-Duhem equation. The treatment is based on combination of the experimental data with the thermodynamic values available on the compressibility factor of pure kerosene at room temperature. It is shown that the activity of TBP in kerosene has a positive deviation from ideality with an activity coefficient derived as follows:1) at X TBP ≤ 0.01: γ TBP = 42.530, 2) at the 0.01 X TBP 0.2: 3) at the higher TBP concentrations 0.2 X TBP 0.97: and 4) at TBP Raoultian concentrations 0.97 ≤ X TBP:γ TBP = 1. These quantities can be utilized at temperature closed to 298 K.

Thermodynamic system which presents the balance of sources and types of

Development and application of the thermodynamic database PRODATA dedicated to the monitoring of mining activities from exploration to remediation - ScienceDirect
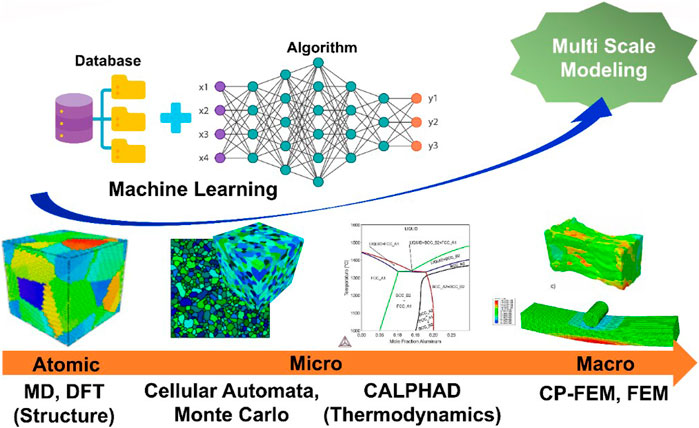
Frontiers Novel Alloy Design Concepts Enabling Enhanced Mechanical Properties of High Entropy Alloys

PDF) Developing a Thermodynamical Method for Prediction of Activity Coefficient of TBP Dissolved in Kerosene

PDF) Machine Learning in Thermodynamics: Prediction of Activity Coefficients by Matrix Completion

Ab initio thermodynamics Max-Planck-Institut für Eisenforschung GmbH

In Silico methods for ADMET prediction of new molecules

Entropy and the Second Law of Thermodynamics: Disorder and the Unavailability of Energy

Water adsorption in the organic phase for the D2EHPA-kerosene/water and aqueous Zn2+, CO2+, Ni2+ sulphate systems
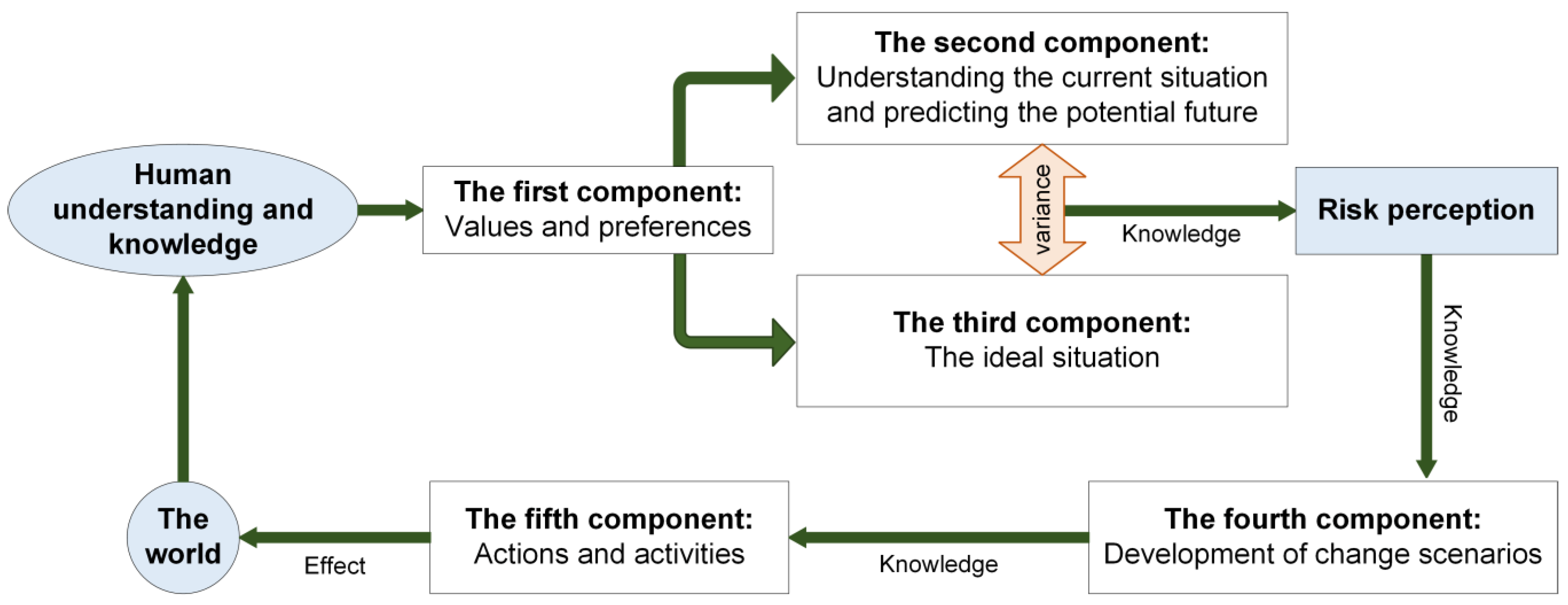
Systems, Free Full-Text







