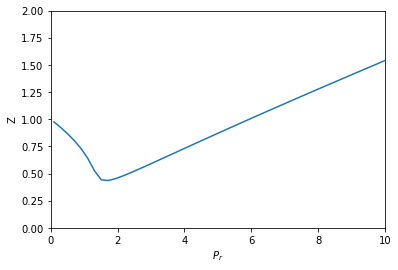
Solved PROBLEM 3. The compressibility factor (Z) for
4.7 (66) · $ 15.99 · In stock

gas laws - How to find the temperature relationship between the isotherms in a compressibility factor (Z) vs pressure graph? - Chemistry Stack Exchange

Compressibility Factor Calculator - File Exchange - MATLAB Central

COMPRESSIBILITY factor Z, Using P and v in 3 Minutes!
Compressibility factor variation from the van der Waals equation by three different approaches

Solved F The compressibility factor ( Z ) of liquid faca
The given graph represent the variation of z compressibility factor z=pv/nRT versis p fpr three real gases A,B,C identify only incorrect statement

Energies, Free Full-Text

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be
SOLVED: PROBLEM 3: The compressibility factor (Z) for nitrogen was measured at -100 °C from 10 to 50 bar pressure. The results are shown below: Pressure (bar) Compressibility factor (Z) 0 14

i.ytimg.com/vi/3ta9OUAC4IY/sddefault.jpg






![PDF] Natural Gas Compressibility Factor Correlation Evaluation for Niger Delta Gas Fields](https://d3i71xaburhd42.cloudfront.net/8eaf43961c0da29108ee5dbc14c9e07b081c25ff/7-Figure2-1.png)