The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate the volume of a 4 mole sample of co, same temperature and pressure (use R = 0.08 L. atm/K.mol (1)
4.8 (397) · $ 9.50 · In stock

Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor z for co at 7c and 100atm is 021 calculate the volume
Click here👆to get an answer to your question ✍️ The compression factor -Z- Co- 7-C and 100 atm is 0-21- Calculate the volume of a 4 mole sample of co- same temperature and pressure -use R - 0-08 L- atm-K-mol -1- 0-192 -2- 0-05 L -3- 0-38 L -4- 0-44 L closed container can be

Modeling Trasport phenomena Part 1 by Alireza Rezayee - Issuu

solution manual for applied petroleum reservoir engineering by craft by kholoud hamad - Issuu

Acentric Factor - an overview
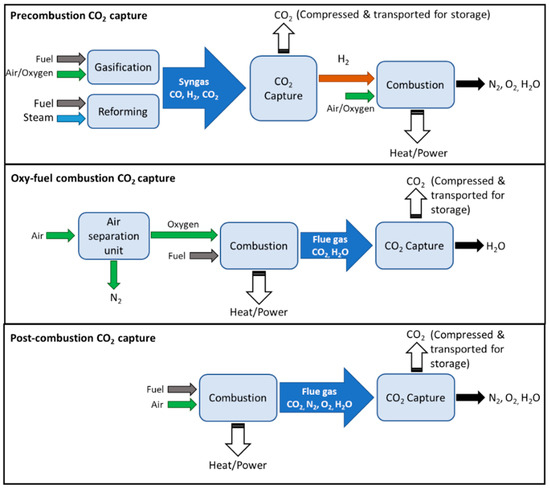
Inorganics, Free Full-Text

Compression Factor Exam Problem using Molar Volumes - Fully Explained!

DEVELOPMENT OF ON-LINE GC/MS MONITORING TECHNIQUES

oxygenated fuel strategies to combat air pollution - Argonne
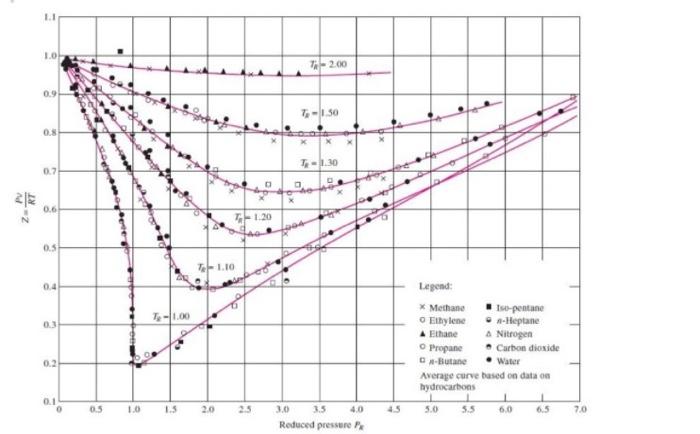
Solved The following figure shows a graph of the compression

Ideal Gas Behavior - an overview

Introduction to Chemical Engineering Thermodynamics Solution Manual - Flipbook by Oya FX Trading & Investments

Answered: (b) A sample of 4.0 mol O, (g) is…

Bioprocess engineering principles by Mauricio - Issuu







