Breaking local symmetry—why water freezes but silica forms a glass
4.8 (536) · $ 22.50 · In stock

Everyone knows that water freezes at 0 degrees C. Life on Earth would be vastly different if this were not so. However, water
Everyone knows that water freezes at 0 degrees C. Life on Earth would be vastly different if this were not so. However, water's cousin, silica, exhibits wayward behavior when cooled that has long puzzled scientists.

Freezing life within refractory, amorphous silicon dioxide
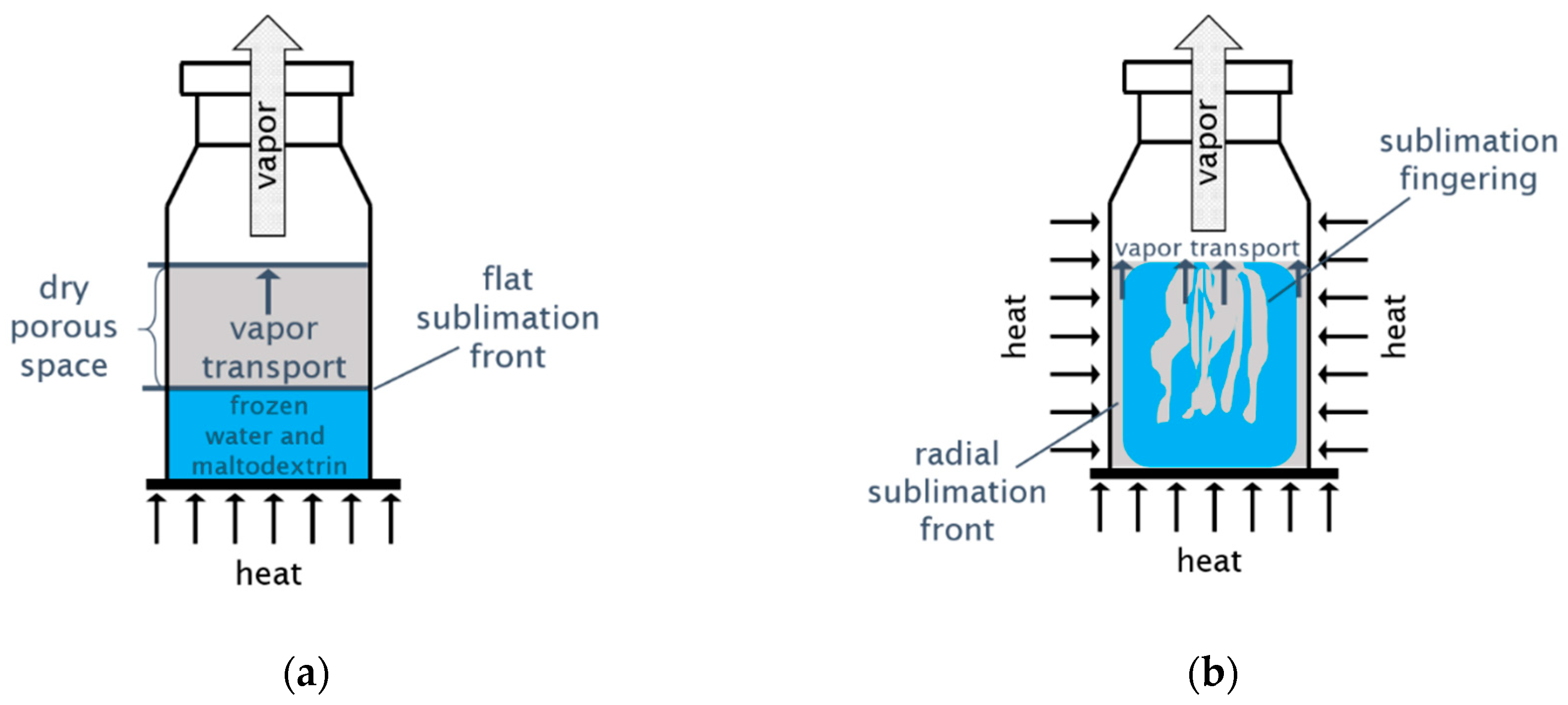
Processes, Free Full-Text

A Journey Through the Gallery of Fluid Motion, Chaosmosis: Assigning Rhythm to the Turbulent
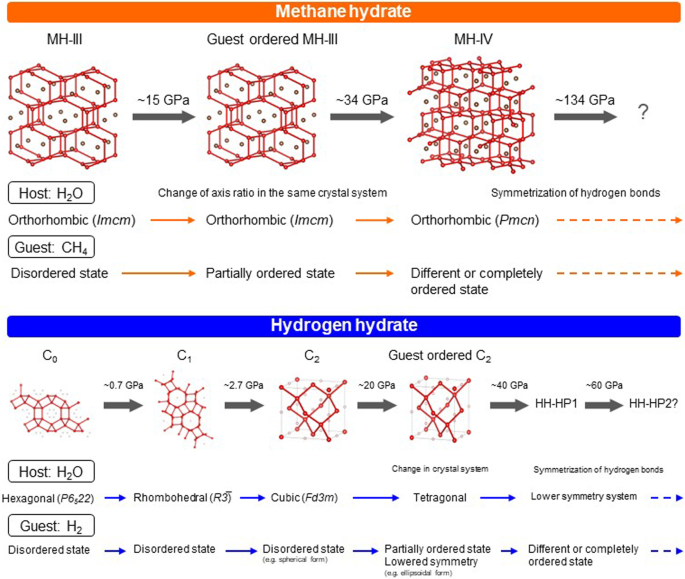
Significance of the high-pressure properties and structural evolution of gas hydrates for inferring the interior of icy bodies, Progress in Earth and Planetary Science
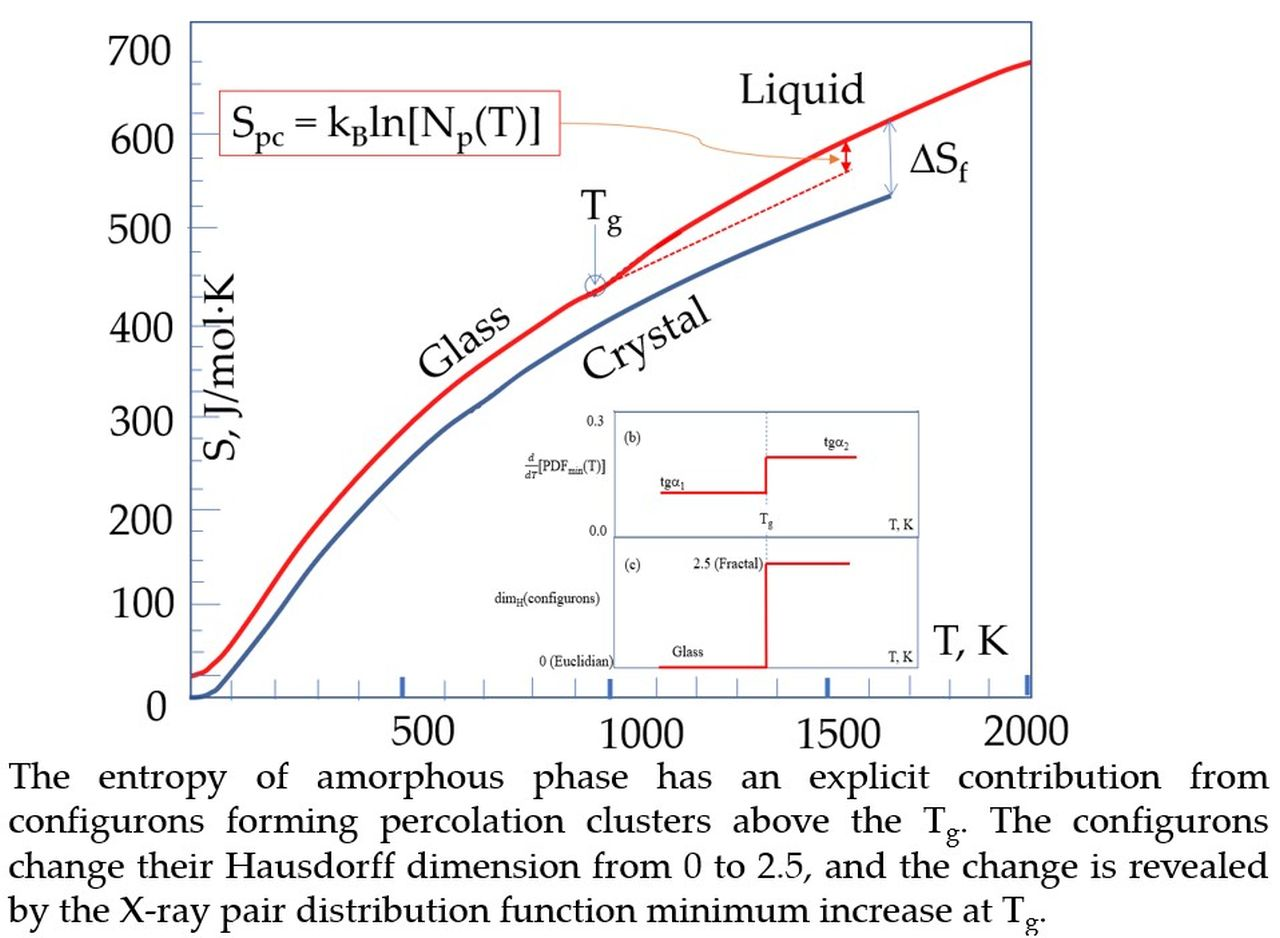
Materials, Free Full-Text
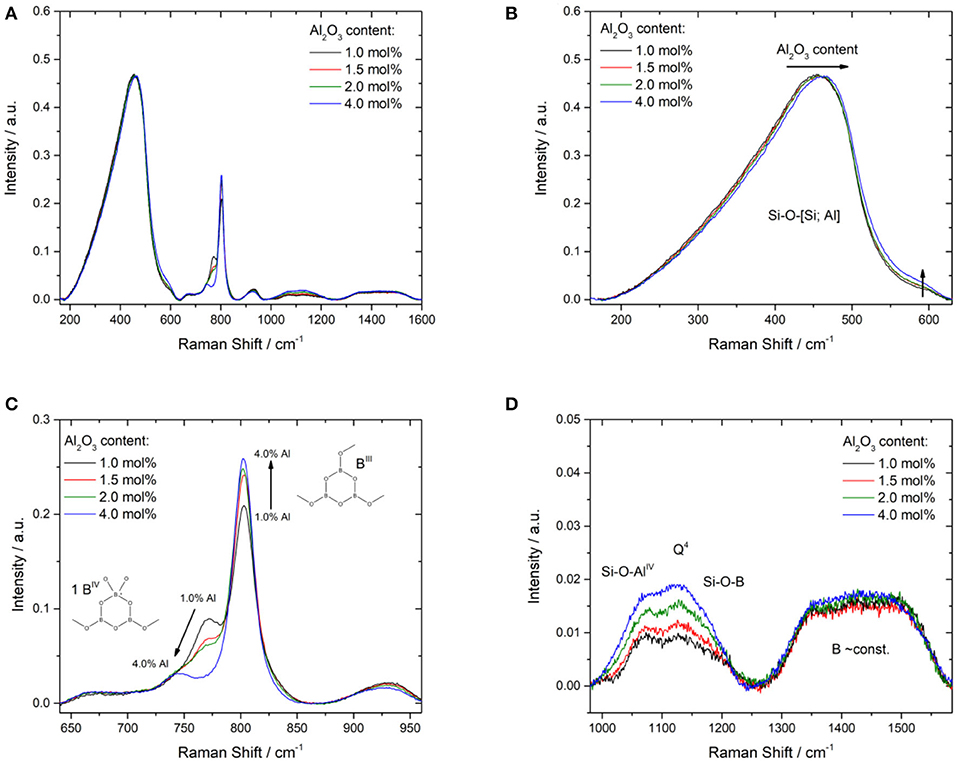
Frontiers Influence of Al2O3 Addition on Structure and Mechanical Properties of Borosilicate Glasses
Dehydration of a crystal hydrate at subglacial temperatures

A day to celebrate chemistry's favorite unit—the mole. But what's

Ice Crystallization in Shear Flows The Journal of Physical Chemistry C

Glass transition - Wikipedia

Materials, Free Full-Text
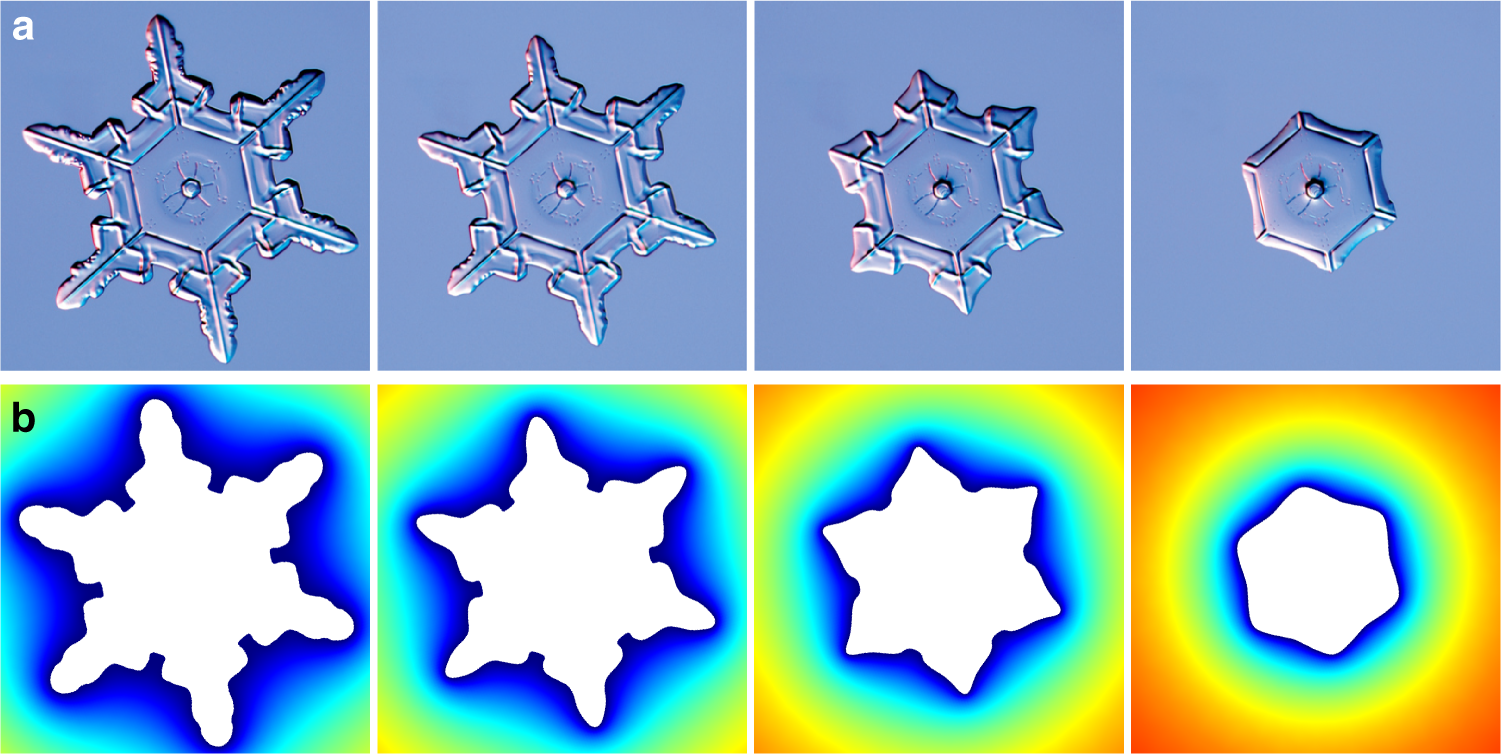
Singular sublimation of ice and snow crystals

Polymers, Free Full-Text
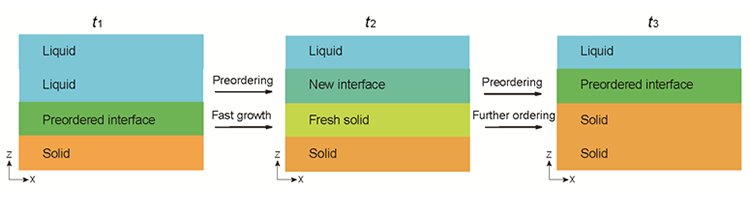
Domino-like crystallization of glass







