The compressibility factor Z a low-pressure range of all gases
5 (726) · $ 5.99 · In stock

Click here:point_up_2:to get an answer to your question :writing_hand:the compressibility factor z at a lowpressure range of all gases except hydrogen is
Click here👆to get an answer to your question ✍️ The compressibility factor Z a low-pressure range of all gases except hydrogen is-Z-1- displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-Pb-RT-Z - - 1 - displaystylefrac-Pb-RT-
The van der Waals equation for real gases is -P-aVm2-Vm-x2212-b-RT

Compressibility factor - Wikipedia
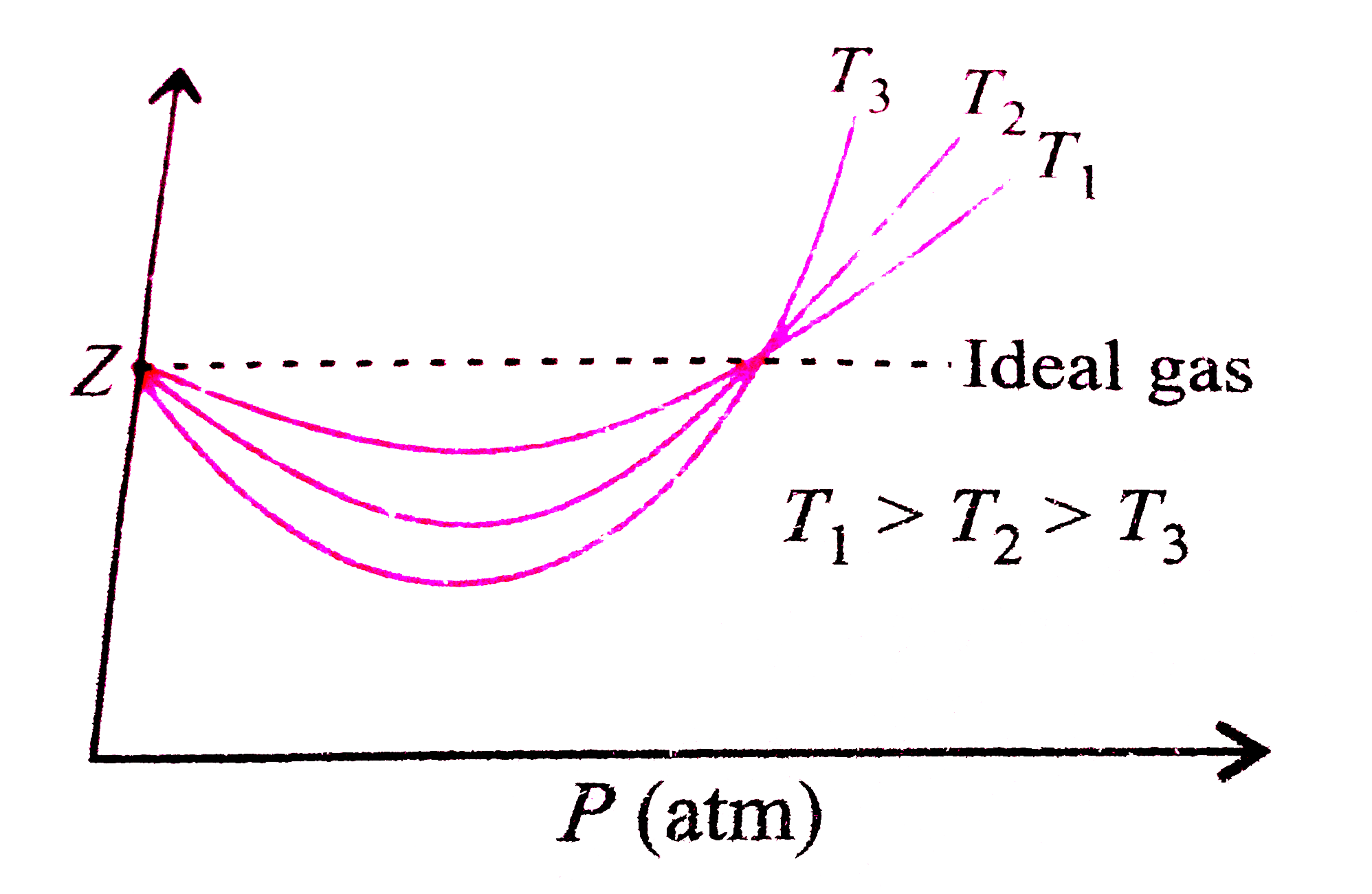
At intermediate pressures , most gases show Z lt 1.

The compressibility factors for 1 mole of real gases at low pressure, high pressure and that of gases of very low molar masses are Z1, Z2 and Z3. These are

4.2: Real Gases (Deviations From Ideal Behavior) - Chemistry LibreTexts

Solved 4 Which of the following statements is incorrect? For

Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt

What is compressibility factor? What is its value for ideal gas

Air Compressibility Factor Table - EnggCyclopedia

Lecture 4-Real-Gases, PDF, Gases
At Critical Temperature,pressure and volume . The compressibility Factor (Z) Is
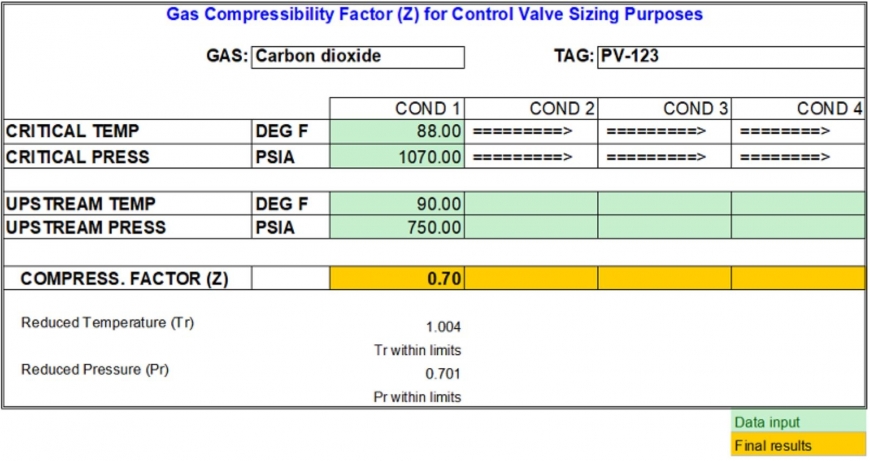
Gas Compressibility Factor and Control Valve Sizing

Torateal gas, the compressibility factor Z has different whues different temperatures and pressures. Which of the following is not correct under the given conditions? (a) Z<1 very low pressure. (b) Z>1 high

Ch2, Lesson E, Page 9 - Generalized Compressibility Chart
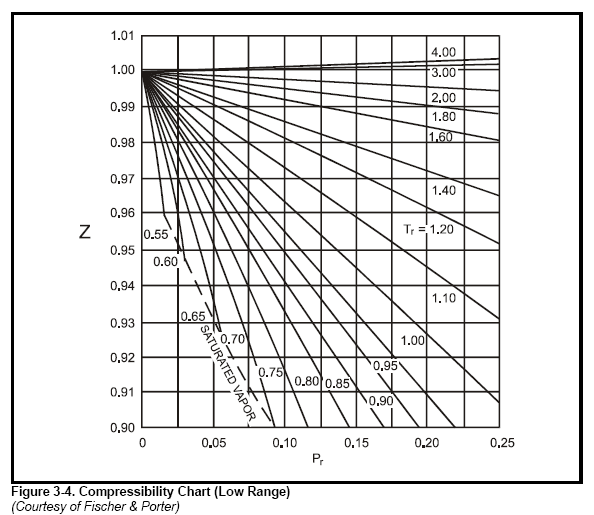
Chapter 3 - Physical Properties of Fluids: Gas Compressibility Factor






