Evaluation and comparison of in vitro intrinsic clearance rates
4.6 (490) · $ 22.00 · In stock


Schematic representation of the strategy used to evaluate the

Evaluation and comparison of in vitro intrinsic clearance rates measured using cryopreserved hepatocytes from humans, rats, and rainbow trout - ScienceDirect

Comparison of the predicted log CLint (LMCI and UMCI) with the

PDF) Quantitative Property-Property Relationship for Screening-Level Prediction of Intrinsic Clearance of Volatile Organic Chemicals in Rats and Its Integration within PBPK Models to Predict Inhalation Pharmacokinetics in Humans

Sherry BLACK, Research CHemist, RTI International, Durham, RTI International, DS

Methods for assessing the bioaccumulation of hydrocarbons and related substances in terrestrial organisms: A critical review - Gobas - 2023 - Integrated Environmental Assessment and Management - Wiley Online Library
![PDF] Assessing Toxicokinetic Uncertainty and Variability in Risk Prioritization.](https://ai2-s2-public.s3.amazonaws.com/figures/2017-08-08/2b5fb29533f54499281920942a4ff0761cb12030/32-Figure3-1.png)
PDF] Assessing Toxicokinetic Uncertainty and Variability in Risk Prioritization.

Evaluation of multigenerational effects of 2‐ethylhexyl 4‐hydroxybenzoate in Japanese medaka - Matten - 2023 - Journal of Applied Toxicology - Wiley Online Library
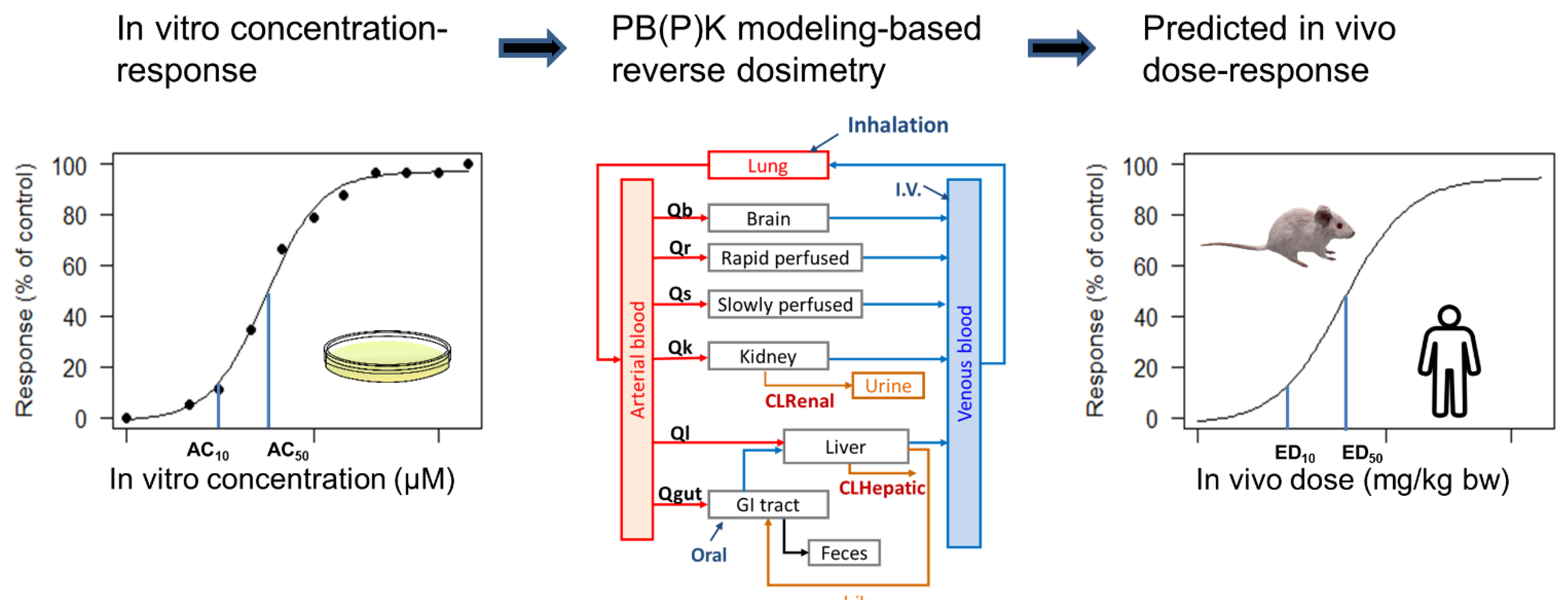
Toxics, Free Full-Text

Correlation of rat and human in vitro pharmacokinetic data. In vitro

PDF) IVIVE: Facilitating the Use of In Vitro Toxicity Data in Risk Assessment and Decision Making

Evaluation and comparison of in vitro intrinsic clearance rates measured using cryopreserved hepatocytes from humans, rats, and rainbow trout - ScienceDirect

PDF) Cryopreserved human hepatocytes in suspension are a convenient high throughput tool for the prediction of metabolic clearance

Correlation of rat and human in vitro pharmacokinetic data. In vitro







