Consider the graph between compressibility factor Z and pressure P
4.7 (758) · $ 12.50 · In stock

Z1 means force of attraction dominating ie a is considerable b can be negligible at low temperature and low pressure Lower is the value of Z easier is the process of liquification
The compressibility factor is actually a factor that corrects the actual value of the gas versus the ideal gas. Let us learn and understand this concept.
Watch this video to understand the behaviour of real gases with the help of the compressibility factor. This is an important topic for JEE main.
What is the compressibility factor, and how does it vary with an increase in temperature and pressure? Watch this video to get the answer. This is an importa
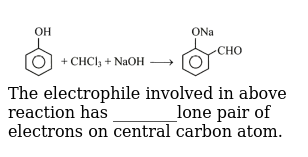
The electrophile involved in above reaction has lone pair of electrons

Compressibility factor z versus 100/V, for several values of Pressure

The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following

A graph Z vs P is plotted N_2 gas different temperatureThe correct relationship between temperatures

PV Compressibility factor Z= nRT is plotted against pressure : N. Ideal gas What is the correct order of liquefiability of the gases shown in the above graph? H

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure
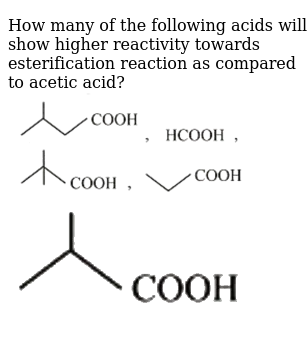
How many of the following acids will show higher reactivity towards es
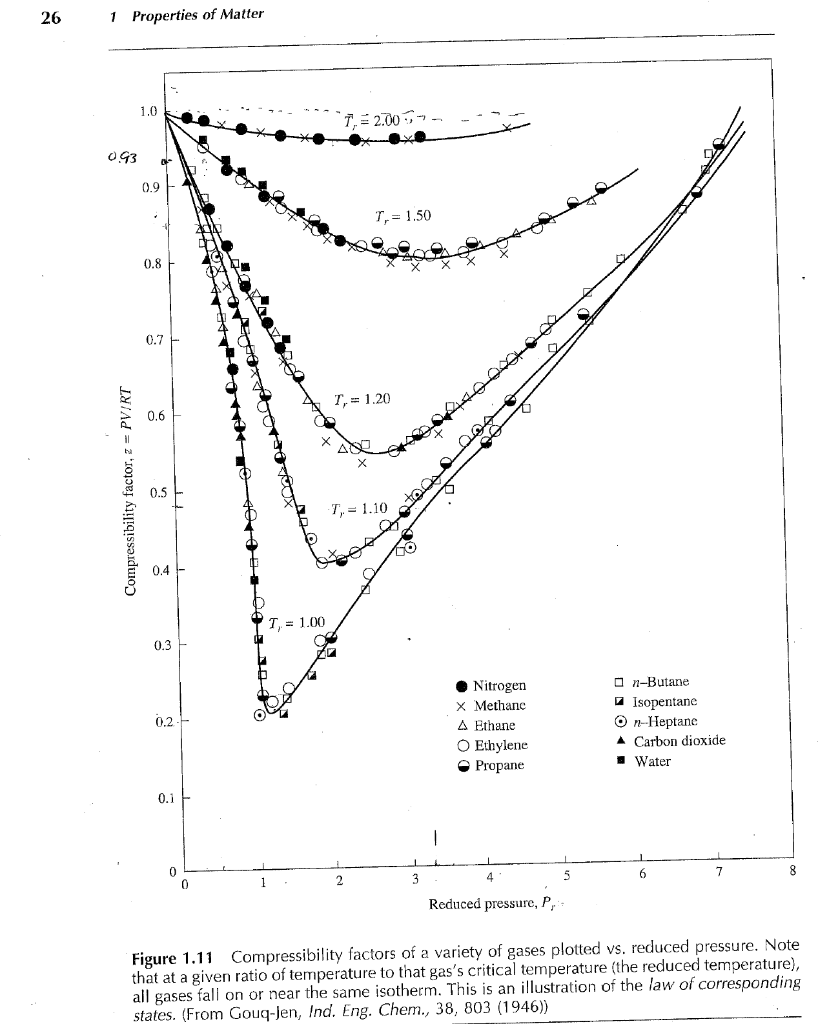
Solved 3) Using the “Z graph” to estimate the compression

Compressibility factor - Wikipedia

Consider the graph between compressibility factor Z and pressure P







